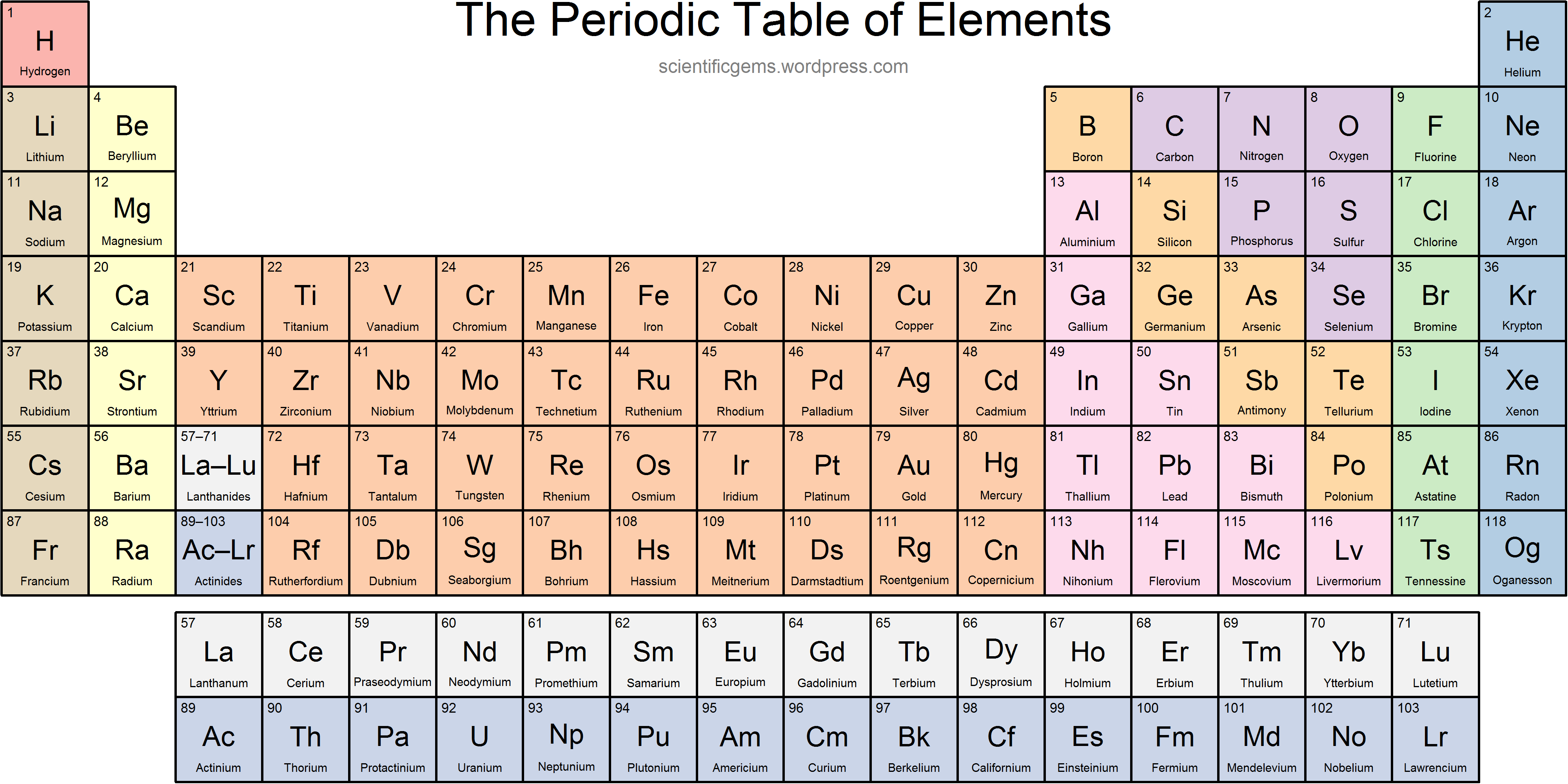
Here the radius of two ions are not equal and few other things need to be discussed before we calculate the ionic radius: Ionic radius is the radius calculated when an atom is bonded with another atom in a molecule by transferring electrons to make ionic bond. The metallic diameter is equal to the distance between two nuclei and the radius is just half of it. The metallic radius can also be calculated by measuring the distance between the two nuclei of the two atoms. The metallic radius is the radius calculated between two metal atoms bonded together in metal cluster. The diameter of an atom is equal to the distance between two nuclei and the atomic radius will be the half of it. When an atom (A) is covalently bonded with another homonuclear atom (A), the atomic radius of that atom can be calculated by measuring the distance between the two nucleus of those two atoms. Covalent radiusĬovalent radius is the radius calculated when an atom is bonded covalently with another atom of same element. There are different methods of calculating the radius by measuring the distance between two nucleus when an atom is bonded in a molecule. So, as the position of electron is not certain rather they are explained as electron cloud around the nucleus, it is hard to measure an atomic radius accurately. Ways to measure the atomic radiusĪccording to the Heisenberg uncertainty principle, it is not possible to measure the momentum and the position of the electron simultaneously. Thus sometime different methods are used to measure the radius while they are bonded in a molecule. As there are no physical existence of orbital in atoms, it is difficult to measure the atomic radius. Send us feedback about these examples.The atomic radius is the size of the atom, typically measured by the distance from the nucleus of the atom to the electron clouds around the nucleus. These examples are programmatically compiled from various online sources to illustrate current usage of the word 'periodic table.' Any opinions expressed in the examples do not represent those of Merriam-Webster or its editors. Jazz Tangcay, Variety, 16 June 2023 See More Sanjana Curtis, Scientific American, 4 July 2023 This idea came from drawing buildings on top of this periodic table and making them into characters. 2023 As the universe expanded and cooled, protons and neutrons were able to assemble into nuclei, producing the lightest elements of the periodic table, and setting the stage for the birth of stars and the synthesis of new elements. 2023 It was declared a success in 2012, and four years later, the International Union of Pure and Applied Chemistry – the world authority on chemistry – officially recognized Element 117 as part of the periodic table, along with three other new elements. Ashraya Gupta, Scientific American, 7 Sep. Thomas Floyd, Washington Post, 16 June 2023 Katie Hafner: Uranium is the last of the naturally occurring elements on the periodic table, with an atomic number of 92. 2023 Utilizing a slew of props - including playing cards, celebrity photos, the periodic table, an audience cellphone and, most memorably, a ripe kiwi - Kwong delivers trick after hypnotizing trick. Jennifer Ouellette, Ars Technica, 29 Mar. 2023 So Smith is in good company with his interactive periodic table project. Chris Impey, Discover Magazine, 27 Oct. 2023 Simple elements in the periodic table have low prices. 
Recent Examples on the Web There’s still a core goal of working out the astrophysical sites where every single element in the periodic table is formed.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
